3 Minute read
A new patented technology with potential to improve outcomes for people undergoing corneal transplant surgery has been developed by polymer researchers at the University of Melbourne, in collaboration with medical specialists.
The need
Millions of people globally suffer from corneal blindness. Partial cornea transplant surgery is the treatment currently delivering the best results for improved vision. However, the procedure is highly challenging, leading to greater uptake of total cornea transplants, despite poorer patient outcomes. Working with medical specialists, researchers at the University of Melbourne have engineered a solution to simplify corneal endothelium transplants and to improve surgical prospects for patients worldwide.
The cornea is the transparent ‘window’ of the eye that covers the iris and pupil, and allows light to enter, and the corneal endothelium is a monolayer of cells at the back of the cornea that acts to maintain corneal clarity.
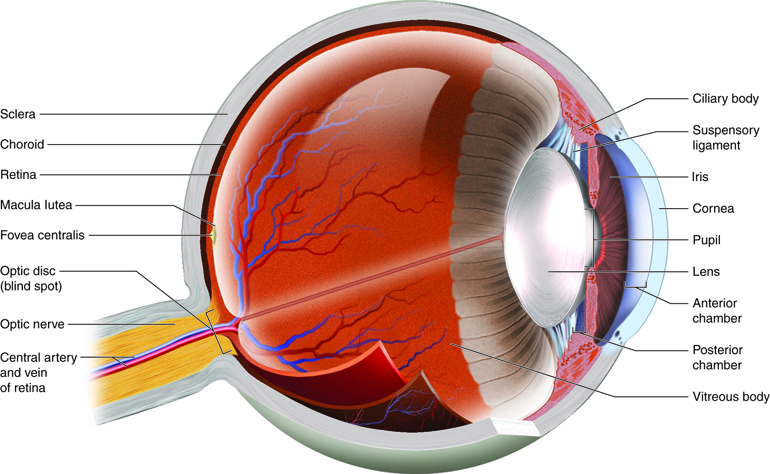
Loss of endothelium function can be caused by ageing, injury or hereditary disease. More than 12.7 million people globally suffer from corneal blindness, making it the fourth most common cause of blindness. Corneal blindness leads to a substantial health and economic burden, often affecting youth from rural areas and lower socio-economic backgrounds. The condition is most common in developing countries. However, many Australians and people from other developed countries also suffer from corneal blindness.
Currently, the best treatment option for corneal endothelial disease is Descemet’s membrane endothelial keratoplasty (DMEK), a partial-thickness cornea transplant procedure. This involves selective removal of the endothelium and its basement membrane (Descemet’s membrane), followed by transplantation of donor corneal endothelium and Descemet’s membrane.
The implanted tissue is only 10-15 microns thick, fragile and has a tendency to scroll, or roll up. The challenge for the surgeon is to unroll and position this delicate graft at the back of the patient’s cornea, a process which alone can take more than two hours. As a result, the majority of surgeons opt for using a simpler yet more intrusive transplant technique, which includes part of the stroma (surrounding cells and tissues), known as Descemet’s stripping automated endothelial keratoplasty (DSAEK). In this case, a much thicker, more robust tissue is implanted. However, the problem with this type of surgery is that the method can lead to poorer visual outcomes and often eventual implant rejection.
Developing the solution
Professor Greg Qiao, Associate Dean (Research Training) in the Faculty of Engineering and Information Technology (FEIT) and leader of the Polymer Research Group, is a global expert in his field of synthetic polymer chemistry, and owner of over 20 patents.
For over a decade, Professor Qiao has been extending his breakthrough development of three-dimensional biomacromolecular scaffolds for soft tissue engineering to thin two-dimensional hydrogels for corneal implants.

Drawing on foundational research over a ten-year period, Professor Qiao and his team have developed a purely synthetic, yet biodegradable, hydrogel scaffold that is capable of supporting a delicate human corneal tissue for eye surgery. The hydrogel is highly transparent and was developed to replicate the strength and thickness of the Descemet’s membrane. In addition, this material facilitates the transport of nutrients, ocular fluids and gases necessary for corneal health, and has been demonstrated to be non-toxic.
Researchers at the University of Melbourne with their collaborators and medical research partners at the Centre for Eye Research Australia (CERA) and Eversight USA have developed a method for sticking this support to the delicate donor corneal endothelium, enabling it to ‘pop-up’ like a tent when implanted into the eye. This technology is now trademarked under the name Hygelix™. The team is nearing 18 months of pre-clinical work to refine and test Hygelix™, with the aim of beginning a first-in-human trial within two years.
The outcome
At present, only one quarter of corneal endothelial transplants in Australia and the United States are performed using the challenging, but more effective, DMEK technique. After Hygelix™ commercialisation, DMEK surgery could become the main corneal transplant method, providing quicker and safer outcomes for patients worldwide.
Banner photo: Getty Images
Eye anatomy diagram: Anatomytool.org (Creative Commons)
Photo of Professor Qiao with Hygelix™: Cesar Nicolas
Partners
Professor Mark Daniell, Head, Corneal Service, Centre for Eye Research Australia
Professor Jean-Pierre Scheerlinck, Faculty of Veterinary and Agricultural Sciences, University of Melbourne
Mr Michael Titus, Vice-President, Clinical Operations, Eversight Vision (USA)
Along with Professor Qiao and his team, these research teams are also part of the BIENCO consortium, which aims to develop a fully bioengineered cornea for the treatment of corneal blindness. Continued work with the corneal gel is essential to BIENCO’s goals.
Funding
Victorian Medical Research Acceleration Fund (VMRAF)
Major reports and publications
Ozcelik, B., Brown, K.D., Blencowe, A., Ladewig, K., Stevens, G.W., Scheerlinck, J.-P.Y., Abberton, K.,
Daniell, M., Qiao, G.G., Biodegradable and Biocompatible Poly(Ethylene Glycol)-based Hydrogel Films for the Regeneration of Corneal Endothelium. Adv. Healthc. Mater. 2014, 3, 1496–1507. DOI: 10.1002/adhm.201400045
Ozcelik, B., Qiao, G.G. and Stevens, G.W., Hydrated Materials: Applications in Biomedicine and the Environment, 2015, p. 71-92, CRC Press.
Hamid, Z.A.A., Blencowe, A., Ozcelik, B., Palmer, J.A., Stevens, G.W., Abberton, K.M., Morrison, W.A.,
Penington, A.J., Qiao, G.G. Epoxy-amine synthesised hydrogel scaffolds for soft-tissue engineering.
Biomaterials 2010, 31, 6454–646 DOI: 10.1016/j.biomaterials.2010.05.008
Daniell, M., Brown, K.D., Gurr, P., Scheerlinck, J.P., Dusting, G., Sawant, O., Titus, M., Qiao, G., Use of novel hydrogel scaffold to assist Descemet’s Membrane Endothelial Keratoplasty (DMEK) surgery. Investigative Ophthalmology & Visual Science, 2020, 61 (7), 3601. DOI:10.1038/s41433-019-0349-7
Patents or IP
Hydrogel preparation and process of manufacture thereof (WO2004055057) (I July 2004). Greg Guang-Hua Qiao, David Henry Solomon.
A. Blencowe, B. Ozcelik, G.G. Qiao, Biodegradable Network Polymers for
Regenerative Medicine and Tissue Engineering, US 2019 / 0194389 A1, (2019).
Re-use this text
Please use the text of this article for your own purposes. The text is licensed under the Creative Commons Attribution (CC BY) 4.0 International license. This lets you copy, transform and share the text without restriction. We appreciate appropriate credit and links back to this website. Other content on this page (such as images, videos and logos) is not covered by the CC BY license and may not be used without permission from the copyright holder. If you have any questions about using this text, please contact the research web team.
First published on 1 September 2022.
Share this article
Related items
-
Tools to improve learning for students with additional needs or disabilities
The SWANs assessment and teaching tools are improving education for children and young people with disabilities or additional learning needs.
-
Lenexa Medical’s new technology to ease pressure for bed-bound patients
A Melbourne-based start-up has developed an innovative product, LenexaCARE, to help prevent pressure injuries by monitoring patient position and posture.
-
Reaching stroke patients in time to give life-saving treatment
Stroke patients in the Mobile Stroke Unit are ten times more likely to be treated in the ‘golden hour’ than patients who go to hospital by ambulance.